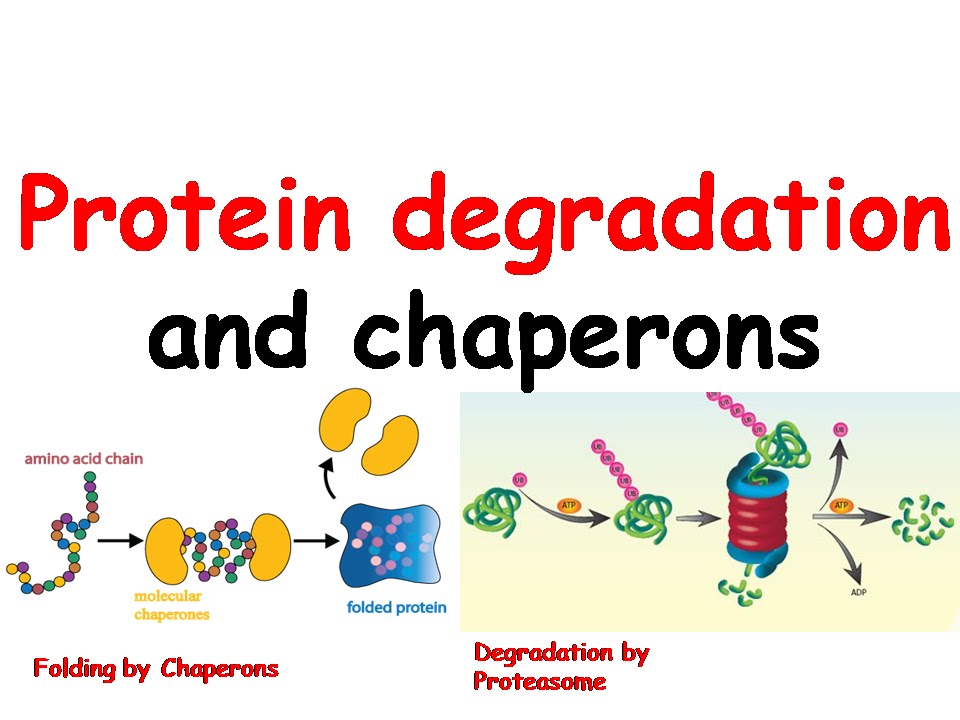 
For example, the accumulation of the misfolded proteins in the cell causes (ER) stress. Hsp 90 chaperones aids in the proper folding of the misfolded proteins in the ER, thereby, it helps to relieve ER stress and its related diseases. They have a positive influence on endoplasmic reticulum (ER) stress-related pathologies. They are termed as chemical chaperons that help to treat several metabolic disorders, such as obesity, type 2 diabetes, and atherosclerosis. These molecules inhibit the signaling pathways involved in the growth and proliferation of tumor cells. Hsp 90 is a type of chaperon which has various therapeutic importance. Both Hsp 70 and 60 have two different forms, where the first form is the binding form and the second form is the folding-active state. A protein that assists the non-covalent folding/unfolding and the assembly/disassembly of other macromolecular structures, but does not occur in these. They have 14 different components with two rings, made up of seven rings. This quarantine mechanism helps to prevent the proteins from getting clumped with other linear polypeptide chains in the cytoplasm. Hsp 60, unlike Hsp 70, which is involved in the prevention of protein aggregation, their function is to isolate unfolded proteins. These Hsp70 proteins recognize the “extended region” with the hydrophobic region of the unfolded protein, and prevent the random aggregation. The hydrolysis of ATP occurring at the N terminal of the chaperon molecule helps in the opening and binding of the substrate to the C terminal. They are monomers with a C and an N domain the C terminal domain binds with the substrate protein, while the N terminal domain contains ATPase. Hsp70 acts as a catalyst in protein folding processes, such as misfolding and refolding of the aggregated proteins, assembling and folding of newly synthesized proteins. The name heat shock protein was first identified from a bacterium that is at high stressful states such as high pH, temperature, and hypoxic conditions. The exposure of the heat destabilizes the protein structure, so during such harsh conditions they need assistance to fold. Thus the concept of molecular chaperones does not contradict the principle of protein self-assembly, but qualifies it by suggesting that in vivo self-assembly requires assistance by other protein molecules.Chaperone molecules are termed as "heat shock" proteins (HSPs), as they are formed in a large amount when the cells get exposed to heat. We study protein quality control across translational models of cancer, cardiovascular disease and neurodegeneration with the goal of defining mechanisms of. Known molecular chaperones do not convey steric information essential for correct assembly, but appear to act by binding to interactive protein surfaces that are transiently exposed during various cellular processes this binding inhibits incorrect interactions that may otherwise produce non-functional structures. The term assembly in this definition embraces not only the folding of newly synthesized polypeptides and any association into oligomers that may occur, but also includes any changes in the degree of either folding or association that may take place when proteins carry out their functions, are transported across membranes, or are repaired or destroyed after stresses such as heat shock. Molecular chaperones are currently defined in functional terms as a class of unrelated families of protein that assist the correct non-covalent assembly of other polypeptide-containing structures in vivo, but which are not components of these assembled structures when they are performing their normal biological functions. 
This introductory article proposes a conceptual framework in which to consider the information that is emerging about the proteins called molecular chaperones, and suggests some definitions that may be useful in this new field of biochemistry.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
